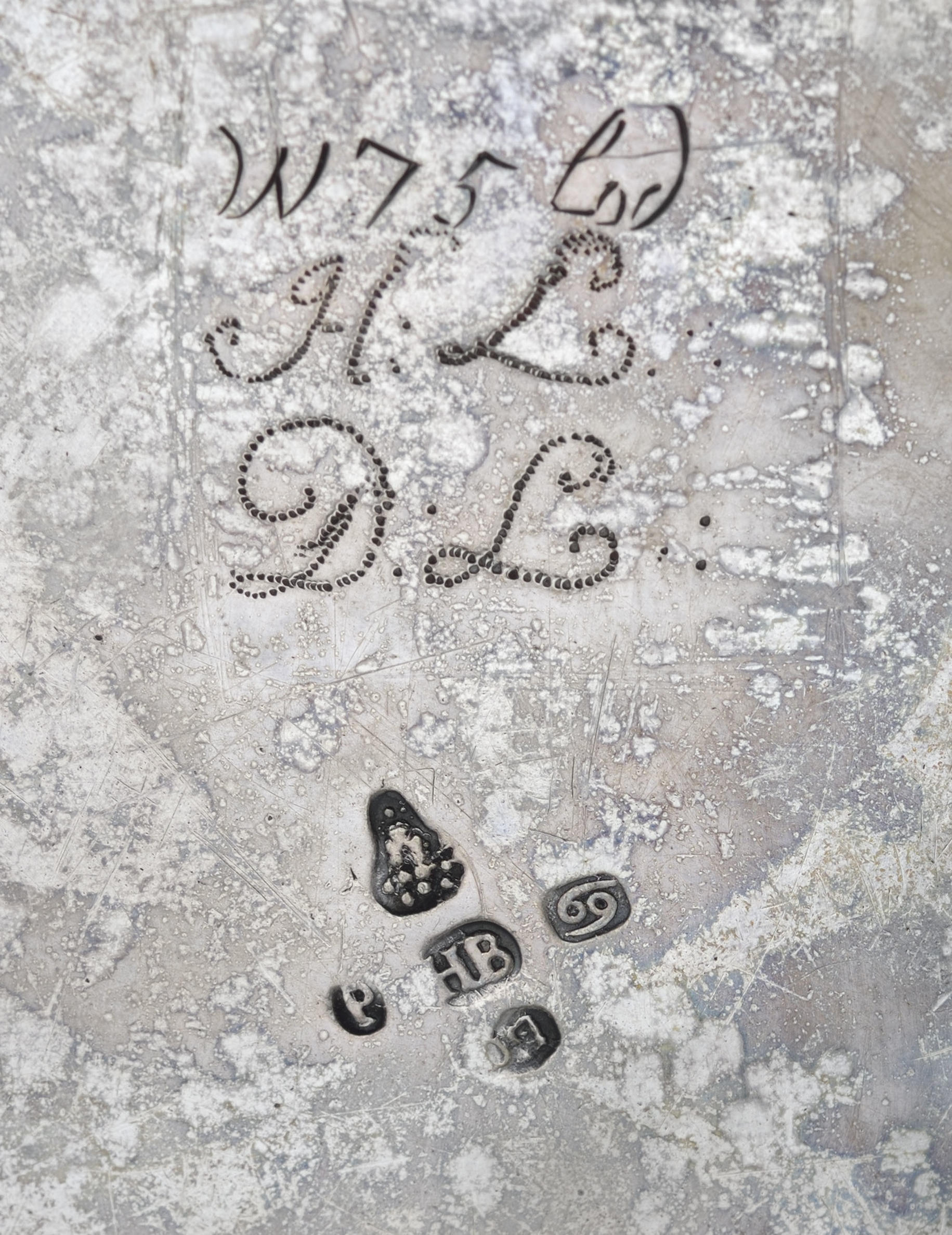When it comes to removing tarnish on objects in the Museum’s collection, there are several options. Our favorite method is to polish silver with a paste we make ourselves by mixing a synthetically prepared calcium carbonate that is purer and more uniform than natural calcium carbonate with deionized water. We work in small areas with a cotton swab dipped in this paste to gradually remove the tarnish and bring back the shine. Calcium carbonate is a gentle abrasive compared to many commercial polishing products, which are often much harsher and leave scratch patterns on the surface. Once the silver is polished, thoroughly rinsed, and dried, we apply a protective lacquer coating to prevent further tarnishing and the need for frequent polishing. Every time a piece gets polished, a tiny amount of silver is removed in the process—therefore, this protective coating helps us avoid polishing more than we have to!